Articles & News on Additive Manufacturing

Case Study: Robotics Integrator Discovers Binder Jet 3D Printing for Automotive End of Arm Tooling
- by Sarah A. Webster
- March 10, 2021
- Case Study
FreeFORM Technologies delivered a complex end-of-arm tooling solution in 17-4PH stainless steel for a high-volume automotive inspection process using binder jet 3D printing technology from ExOne.

Voxel8 Collaborates with Eddy Ricami to Bring Advanced Additive Manufacturing Tech to High Fashion Industry
- March 9, 2021
- News
Voxel8's transformative multi-material ActiveLab® system will enable digital manufacturing of unique designs in footwear and accessories for top fashion brands.

igus Linear Axes Manufacture Spare Parts in Space
- March 4, 2021
- News
Germany students develop 3D printer for cost-effective production of structural elements.

Siemens and EOS North America Partner to Improve AM Efficiency and Profitability
- February 23, 2021
- News
EOS North America to integrate and resell Siemens’ additive manufacturing (AM) software into its 3D printing systems.

Intellegens and Ansys to Empower Additive Manufacturing with Material Intelligence Through Machine Learning
- February 17, 2021
- News
Machine learning specialist Intellegens and engineering simulation leader Ansys have announced a collaboration to integrate machine learning methods into Additive Manufacturing (AM) workflows, accelerating the development of reliable and repeatable AM processes.

Despite A Global Pandemic, 2020 Unlocked Unexpected, Game-Changing And Inspirational Uses of 3D Printed Designs
- December 18, 2020
- News
Ultimaker, the global leader in professional 3D printing, today launched its inaugural Innovators List: a celebration of the most unexpected, game-changing and inspirational uses of 3D printing across business and education this year.

Advantech & Quanergy Announce Partnership to Build LiDAR-Enabled 3D IoT Solutions for Smart City & Logistics
- October 21, 2020
- News
Advantech Service-IoT GmbH announces its partnership with Quanergy systems Inc., the leading provider of LiDAR (Light Detection and Ranging) sensors and smart perception solutions.

Lifting More Than You Can Handle?: How to prepare for an unexpected spike in demand
- by Jonathan Wilkins, EU Automation
- July 6, 2020
- Opinion
By Jonathan Wilkins
This article explains how the manufacturing industry can prepare for unanticipated periods of increased demand.

Pepperl+Fuchs announces 2020 Online Summit
- June 24, 2020
- News
Pepperl+Fuchs’ 2020 Online Summit is a free digital platform that will allow customers and other interested parties to learn about the latest trends and solutions in factory and process automation—even during the COVID-19 pandemic.

Saratech announces acquisition of Apex Digital Manufacturing Solutions
- June 9, 2020
- News
Saratech will take over all of Apex's PLM software license and subscription sales, support services, and training classes.

ANCA starts manufacturing ventilators for Australian COVID-19 fight
- May 21, 2020
- News
This initiative directly addresses the critical supply of mechanical ventilation equipment to support COVID-19 patients requiring ventilation to survive. Banding together with other Australian manufacturing businesses, the consortium can harness various industry capabilities to make more ventilators sooner.

Industrial Internet Consortium (IIC) announces Contilio as winner of Smart Construction Challenge
- April 15, 2020
- News
As the winner, Contilio was awarded €25,000 and the opportunity to deliver a live proof of concept (POC) at the TÜV SÜD International Business Park in Singapore.

Eaton uses 3D printing to deliver face shields to hospitals in four states
- April 9, 2020
- News
The first batch of face shields made at Eaton’s Additive Manufacturing Center of Excellence in Southfield, Michigan were donated to hospitals in Ohio, New York, New Jersey and Michigan.

CoVent-19 Challenge Asks Millions of Designers and Engineers to Respond to the Ventilator Crisis
- April 2, 2020
- News
The CoVent-19 Challenge is open to teams and individuals anywhere, and finalists will work directly with Stratasys 3D printing experts and the CoVent-19 Challenge team to turn their designs into prototypes for testing.

Siemens makes additive manufacturing capabilities, 3D printers available to medical community
- March 27, 2020
- News
The Siemens AM network is available globally and covers the entire value chain – from upload and simulation to checking the design up to the printing process and associated services.
AddUp and Oak ORNL announce collaboration to advance metal additive manufacturing tooling applicatio
- January 6, 2020
- News
A $2.7 million cooperative research and development agreement (CRADA) between AddUp Group, a global machine and solutions provider for metal additive manufacturing (AM), and the US Department of Energy’s (DOE) Oak Ridge National Laboratory (ORNL) aims to advance laser powder bed fusion (LPBF) technology for the manufacturing of metal molds with complex geometries for the tooling industry.
Siemens announces launch of Additive Manufacturing (AM) Network
- November 21, 2019
- News
Siemens’ AM Network is designed for enterprises, suppliers and partners that are looking to accelerate the adoption of AM for industrial processes and applications.
How Viable is Additive Manufacturing?
- October 17, 2019
- Feature
By Mark Howard, EU Automation Additive manufacturing (AM) is drastically altering a range of industries, from manufacturing to the medical sector. But what are the limitations of the technology? This article shares some insights behind the manufacturing and legal issues surrounding additive manufacturing.
Senvol announced President Zach Simkin election as Chairman for SME’s Additive Manufacturing (AM)
- August 7, 2019
- News
The primary responsibility of the SME AM Advisory Committee is to advise SME on initiatives, activities, policy, and strategy related to AM.
The Need for Green in 3D Printing
- July 29, 2019
- Feature
By Ben Smye, Matmatch It has been 35 years since the first instance of what we now think of as additive manufacturing (AM), or 3D printing. Lauded for its rapid production speed and minimal waste, the process is becoming a staple of product prototyping. However, there is still room to improve the sustainability of this process.

IMTS is the Additive Manufacturing Destination for 2022
- October 29, 2021
- News
IMTS 2022 will showcase AM solutions as part integrated manufacturing technology offerings in the Metal Removal, Quality Assurance, CAD/CAM, and other Technology Pavilions covering 1.1 million sq. ft. of exhibit space.

CGTech Highlights VERICUT 9.2 Simulation, Composites and Additive Capabilities at CAMX
- September 16, 2021
- News
CGTech will demonstrate VERICUT 9.2 at the Composites and Advanced Materials Expo (CAMX) from Oct. 19-21 in booth W47.

CGTech to Demonstrate VERICUT 9.2 at CMTS
- September 14, 2021
- News
CGTECH will exhibit VERICUT 9.2 in booth #2507 at the Canadian Manufacturing Technology Show (CMTS) from Oct. 4-7 at the International Centre in Toronto, Ontario. VERICUT is the industry standard for CNC machine simulation to detect incorrect tool path motions, collisions, bugs and other areas for improvement.

CGTech to Demonstrate VERICUT 9.2 at WMTS 2021
- August 26, 2021
- News
CGTech will feature VERICUT version 9.2 in booth #334 at the Western Manufacturing Technology Show (WMTS).

Results of Shapeways ‘State of 3D Printing’ Survey Show Significant Shift to Digital Manufacturing
- August 24, 2021
- News
3D Printing now mainstream among 75% of mid-sized manufacturers polled, yet more flexibility needed to meet demands for quality, personalization and sustainability.

3D Printing Cooperation: Photocentric and CoreTechnologie Enter into Partnership
- June 25, 2021
- News
CoreTechnologie has developed a tailored version of its 3D printing software specifically for the requirements of Photocentric 3D printing machines and processes. In future, the tool will be delivered together with the printers of the British printing machine manufacturer.
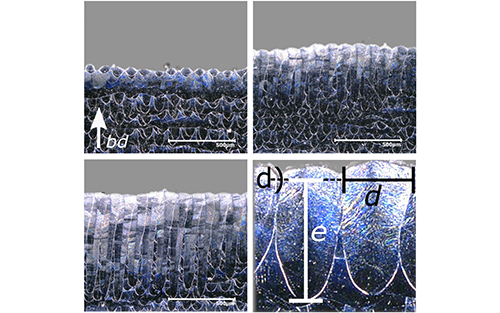
Mikrotron Cameras Help Researchers Create Alternative Method to Monitoring Additive Manufacturing
- May 25, 2021
- News
Non-destructive quality system measures melt-pool width and depth.

Hexagon’s Manufacturing Intelligence Division Thought Leaders Speak at 2021 Additive Manufacturing Users Group
- May 4, 2021
- News
AMUG Programming includes three Hexagon experts covering additive manufacturing workflow, 3D scanning, inspection and process optimization.

Global Challenges Drive Double Digit Growth in Additive Manufacturing Through 2025
- March 30, 2021
- News
New ARC Advisory Group research on the global Additive Manufacturing market reveals that the intersection of 3D printer innovation, software advances, and material creation with the COVID-19 pandemic is shining a new light on industrialized additive manufacturing.

3D Printing Startup Fortify Secures $20M in Series B Funding Led by Cota Capital
- March 18, 2021
- News
Funds will be used to scale up manufacturing, the team, and materials development programs.
Moving Beyond Prototypes: Is 3D printing the answer for the manufacture of safety-critical vehicle p
- July 24, 2019
- Feature
By Kostas Poulios, Pailton Engineering This article explains how 3D printing is used in large vehicle manufacturing and whether this method could play a role in the production of safety critical parts.
Siemens to help European Space Agency develop aerospace design applications for metal additive manuf
- June 20, 2019
- News
The Design4AM project will result in a validated process for using Siemens’ additive manufacturing software to design and produce optimized, light-weighted structural parts for space applications, such as, among others, fittings (structurally bonded to CFRP panel), supports, and bipods.
Sandvik Additive Manufacturing announces creation of 3D printed diamond composite
- May 24, 2019
- Case Study
The diamond composite will be unveiled at the RAPID + TCT show in Detroit May 21 - 23, 2019, North America’s event for Additive Manufacturing.
Sandvik creates 'unsmashable guitar' via additive manufacturing, 3D printing for Swedish guitarist
- April 11, 2019
- Case Study
Global engineering company Sandvik decided to test its additive manufacturing and 3D printing techniques by building the world’s first all-metal, unbreakable guitar and letting Malmsteen unleash his smashing skills.
Carnegie Mellon research aims at identifying causes for defects in 3D Printing
- February 22, 2019
- News
New research published today in Science, led by researchers from Carnegie Mellon University and Argonne National Laboratory, has identified how and when these gas pockets form, as well as a methodology to predict their formation – a pivotal discovery that could dramatically improve the 3D printing process.

Evaluation of Different Additive Manufacturing Processes with Liquids and Pastes
- by Viscotec
- February 19, 2019
- News
The use of additive manufacturing processes is increasing within industrial applications, as they bring some significant customer benefits compared to conventional processes.
Evaluation of Different Additive Manufacturing Processes with Liquids and Pastes
- February 18, 2019
- News
By Viscotec The use of additive manufacturing processes is increasing within industrial applications, as they bring some significant customer benefits compared to conventional processes.
Mitsubishi Electric robot helps English university explore 3D printing and art
- July 27, 2018
- Case Study
Additive Manufacturing (3D printing) is increasingly being used in the manufacturing industry for prototyping, low volume manufacture and for making items with complex shapes which cannot easily be reproduced by other means.
Is the Medical Device Regulation a Threat to Additive Manufacturing?
- May 23, 2018
- Feature
By Bryan Austin, Renishaw the European Union’s (EU’s) new Medical Device Regulation (MDR) could drastically impact the way that medical devices are made in the EU, particularly those that are produced using additive manufacturing (AM). This article provides advice on how companies in the additive manufacturing industry should handle the MDR.
Renishaw announces launch of additive manufacturing guide
- May 8, 2018
- News
The company will use the information hub to educate and inform customers and the engineering community using videos, case studies, feature articles, industry news and opinion pieces.

Quickparts Announces Two Texas Roadshows in December
- December 4, 2023
- News
Quickparts, a leading manufacturer providing custom prototypes and low-volume production parts, is excited to announce its upcoming Texas Roadshows, taking place in Austin and Dallas on Dec. 5 and 6, respectively.

Ricoh and Siemens Collaborate to Realize the Industrial Aluminum Binder Jetting Process for Mass Production
- October 31, 2023
- News
Ricoh is leveraging Siemens’ Additive Manufacturing Network capabilities to maximize the efficiency of the process and to achieve the scale required to take advantage of BJT in an industrial setting.

Beyond the Surface: The Power of Density Determination in Additive Manufacturing
- by Philipp Pruesse
- October 17, 2023
- Feature
In this article, we will delve deep into the significance of density determination in AM, exploring how it assesses the quality of metal 3D printed parts and the impact it has on various industries.

SmarTech Analysis Reports 3D Printing Markets at $3.52B in Q2 2023
- October 10, 2023
- News
The global additive manufacturing (AM) market recorded less than 1% estimated sequential growth in Q2 2023 versus Q1 2023, due in part to mounting economic challenges affecting hardware investments.

3D Printing Industry Totals $3.7B in Q1 2023 with 10% Sequential Growth
- June 22, 2023
- News
The first quarter of 2023 in the global additive manufacturing market recorded double digit sequential growth versus Q4 2022, despite mounting economic challenges.

Mikrotron Camera Helps Scientists Devise Better Quality Control Method for Additive Manufacturing
- May 12, 2023
- News
Scientists at the Technical University of Darmstadt have developed a highly accurate method for determining thermal solidification characteristics of metal parts built with laser-based powder bed fusion, an additive manufacturing technique.

KUKA Highlights New Automation in Mobility, Education, Safety and Additive at AUTOMATE 2023
- April 27, 2023
- News
During AUTOMATE 2023, KUKA Robotics will feature a well-rounded range of solutions for meeting the automation mobility, education, safety and other needs of today’s manufacturers.

Wayland Additive Raises Money to Meet Demands of Growing Global Customers Base
- April 20, 2023
- News
Wayland Additive Limited has announced the close of a £4.6 million funding round, with backing from existing investors Longwall Ventures, Parkwalk Advisors and ACF Investors.

Are You Ready for the Future of Composites Manufacturing?
- April 4, 2023
- News
At JEC World 2023, held April 25-27 at the Paris Nord Villepinte Exhibition Centre, CGTech will demonstrate how advanced programming strategies and simulation can lead to the production of better composite parts.

Wohlers Report 2023 Unveils Continued Double-Digit Growth
- April 3, 2023
- News
Wohlers Report 2023 shows an overall worldwide growth in AM products and services of 18.3%, continuing a trend of double-digit AM industry revenue growth in 25 of the past 34 years.

Nikon Announces Investment in Hybrid Manufacturing Technologies Global, Inc.
- September 14, 2022
- News
Nikon Corporation (Nikon) has announced an investment in Hybrid Manufacturing Technologies Global, Inc.

Ultimaker and MakerBot Announce Closing of Merger
- September 13, 2022
- News
The new private company will focus on accelerating 3D printing innovation and global adoption of desktop 3D printing solutions.

igus' Custom Maintenance-free Toothed Belt Pulleys Delivered in 48 Hours
- July 27, 2022
- News
igus' reduces delivery time and makes online ordering of custom toothed belt pulleys particularly easy with its latest 3D-print offering.

Siemens Opens Additive Manufacturing Hub in Charlotte to Accelerate Industrial Digitalization
- July 25, 2022
- News
Siemens announces the opening of its Center for Advanced Technology Collaboration Hub (CATCH), located in Charlotte, N.C.

Growth of 3D Printing Could Be Derailed by Supply Chain Difficulties
- July 14, 2022
- News
Latest insight reports associated with the recently released SmarTech data indicate that the looming macroeconomic challenges could derail the fast pace of growth observed at the start of the year.

Compact Motion Control Helps Keep 3D Printing Equipment in Line with End User Requirements
- by Paxton Shantz
- June 2, 2022
- Feature
The success of additive manufacturing technology is often measured by the efficiency and reliability of the controls.

Sciaky's Electron Beam Additive Manufacturing Technology Introduced to President Biden
- May 18, 2022
- News
Sciaky, Inc. announced that its Electron Beam Additive Manufacturing (EBAM) technology was introduced to President Biden during an AM Forward address at United Performance Metals in Hamilton, Ohio on May 6, 2022.

ABB Robotics Showcases Future of Retail Using Recovered Marine Plastic at London’s Selfridges
- April 8, 2022
- News
ABB robot 3D additive printing will use upcycled Parley Ocean Plastic to create range of designer items.

Siemens Enables You Mawo and Additive Scale to Scale Up Additive Manufacturing of Bespoke Eyewear
- November 19, 2021
- News
The carbon footprint will be reduced by up to 58% with additively manufactured eyewear.

Siemens’ AM Network to Facilitate Additive Manufacturing Workflow and Collaboration at Schaeffler
- November 12, 2021
- News
Siemens Digital Industries Software announced that its Additive Manufacturing Network platform has been implemented at Schaeffler to connect its community of design engineers with its AM Fab Shop for additive manufacturing to facilitate collaboration, speed time to production and to foster greater application of the benefits of additive manufacturing (AM) within its value-chain.

Additive Manufacturing Industry Grew 23% in 2022 to $13.5B
- March 21, 2023
- News
The fourth quarter of 2022 capped a strong year for additive manufacturing markets, according to SmarTech Analysis.

Jabil Forecasts Continued Growth as Additive Manufacturing Market Matures
- January 11, 2023
- News
Jabil Inc. offers a forecast of five important additive manufacturing trends that will continue to accelerate innovations in the new year.

NexTech AR Solutions Receives Patent Protection on Artificial Intelligence 3D Model Creation
- January 11, 2023
- News
NexTech AR Solutions CEO Evan Gappelberg told Proactive that the company has been granted a patent for its artificial intelligence technology 3D model creation.

Festo Introduces Online 3D CAD Configurator for Actuators and Accessories
- January 11, 2023
- News
A 3D CAD file of the actuator assembly is ready for download at the end of configurator’s streamlined design process.

New Additive Manufacturing Service Set to Disrupt Flow Control MRO Market
- January 6, 2023
- News
Additive manufacturing has the potential to revolutionize in-field control valve maintenance, potentially saving process and industrial plants millions of dollars a year in maintenance, repair and operation (MRO) costs.

Hexagon and Raytheon Technologies Developing Simulation Tool To Improve 3D Printing Build Quality
- November 2, 2022
- News
Hexagon and Raytheon Technologies partner to deliver a simulation tool that will enable evaluation and optimization of metal additive manufacturing processes through the prediction of thermal history and defects at the laser path and powder layer scale.

Siemens and Desktop Metal Join Forces to Accelerate Sustainable Additive Manufacturing at Scale
- October 11, 2022
- News
Siemens and Desktop Metal, Inc., global leaders in digitalization and additive manufacturing technologies, announced a multi-faceted partnership aimed at accelerating the adoption of additive manufacturing for production applications with a focus on the world’s largest manufacturers.

Market for Additive Manufacturing Grows Despite Macroeconomic Headwinds
- October 11, 2022
- News
ARC’s new market research report on additive manufacturing shows that the AM ecosystem has reached a pivotal point of opportunity with the crisscrossing of macroeconomic headwinds and advancement of technologies.

Additive Manufacturing Markets Totaled $3B for Second Consecutive Quarter
- October 3, 2022
- News
The second calendar quarter of 2022 saw continuing year over year growth for the additive manufacturing market, coming in 27% higher compared to the same period in 2021.

Rivelin Robotics to Introduce Netshape Robots for Metal AM Post-processing at Formnext 2022
- September 27, 2022
- News
Rivelin Robotics is pleased to announce that it will be exhibiting at Formnext in Frankfurt Nov. 15-18, 2022 at Stand B41 in Hall 12.
Subscribe to our publications
- eBooks
- Newsletter
Receive the latest automation content, sent to your inbox in the form of enewsletters or ebooks. You pick the topics, we take care of the rest!
Subscribe

