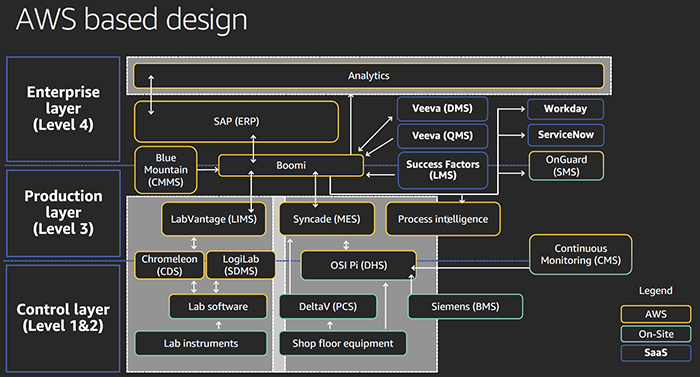
Moderna’s award-winning cGMP clinical development manufacturing facility—based in Norwood, Mass. —has a native digital process automation design built on Amazon Web Services (AWS). It serves as a great example of the integration of an entire manufacturing business using the latest technological building blocks, architecture and IoT ecosystem. It’s fitting for Moderna to bring us this fresh design because the company’s culture is “bold, relentless, collaborative and curious.” Moderna’s facility won the 2019 International Society for Pharmaceutical Engineering (ISPE) Facility of the Year Award .
“In 2019, the facility was named Facility of the year by the international Society for Pharmaceutical Engineering (ISPE),” said Roland Smith, Sr. Director, Digital GxP Systems Moderna. “What differentiated the site, according to ISPE, was fully-integrated Native Digital Manufacturing capabilities built on AWS .” The Moderna native digital process automation design eliminates the large divide between business systems and production that in the past meant disjointed and unsynchronized operations , resulting in inefficiencies from supply chain through to customers. Major goals achieved include:
-
Better data - better decisions
-
Innovation - within existing workflow
-
Prediction - identifying the process variables that are related to an outcome
-
Better real-time process modeling, simulation, and visibility
-
Maximized operator performance
|
|
|
Roland Smith, Sr. Director, Digital GxP Systems Moderna described the architecture at the AWS re:Invent 2019 event. |
About Facility of the Year Awards (FOYA)
Established in 2004, The Facility of the Year Awards (FOYA) recognize state-of-the-art projects utilizing new, innovative technologies to improve the quality of products, reduce the cost of producing high-quality medicines ,and demonstrate advances in project delivery. The FOYA program provides a platform for the pharmaceutical science and manufacturing industry to showcase its accomplishments in facility design, construction , and operation, while sharing the development of new applications of technology and cutting-edge approaches. The International Society for Pharmaceutical Engineering (ISPE) is the world’s largest not-for-profit association serving its members through leading scientific, technical , and regulatory advancement across the entire pharmaceutical lifecycle.
The 18,500 members of ISPE are building solutions in the development and manufacture of safe, effective pharmaceutical and biologic medicines, and medical delivery devices in more than 90 countries around the world. Founded in 1980, ISPE has its worldwide headquarters and training center in North Bethesda, Maryland USA, and its operations center in Tampa, Florida USA. Visit www.ISPE.org for more information.
Key success factors
Roland Smith, Sr. Director, Digital GxP Systems Moderna , noted key factors for success in his presentation at the Amazon AWS re:Invent 2019:
-
Top management's support of digital commitmentreally makes the discussion of digital at Moderna not an "if" but a "when." Removing those headwinds allows us to move faster in our digital transformation at a lower cost.
-
A unique operating model removes organizational silos. For example, my group owns digital, shop floor automation, and analytics. That allows us to rapidly improve as we see opportunities to streamline or make data more consistent.
-
We strongly believe the user experience and lean principles go hand in hand. if you make an operator’s job easier , the process will inherently be more efficient.
-
We are very data-driven , believing the value of collecting data is key to our future success.
-
Designed a future proof ecosystem built on AWS.
-
There is no member of my team who is not currently working on at least one side innovation project that will disrupt how they will normally work.
-
From the start , we have tried to integrate all of our systems to eliminate non-value added manual transcription and improve data consistency. Currently , we have more than 200 integrations across our digital systems.
-
We have more than 40 integrated robots and 100 integrated pieces of process equipment. Analytics and learning are built into everything. Integrating data into everything accelerates insight and improves data quality accelerating insights.
Bringing cloud to operations
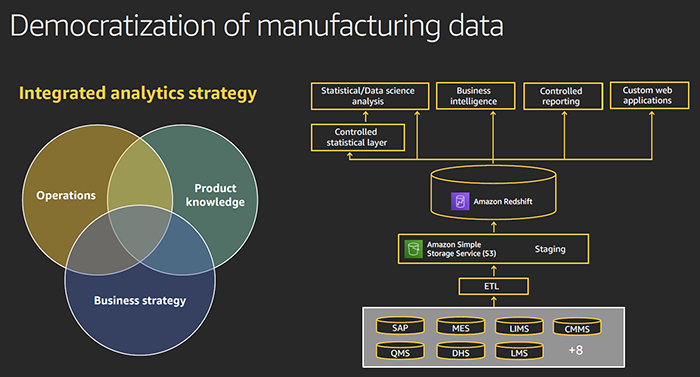
Smith discussed how many of his peers are trying to take operations to the cloud , but at Moderna they are bringing the cloud to operations. Smith noted about 95% their digital landscape computing capacity resides on the cloud, primarily on AWS. Data is housed in Amazon Redshift cloud data warehouse or what some would term a “Data Lake."
|
|
|
Flexible data strategy separated into three segments. Data is housed in Amazon Redshift cloud data warehouse. |
Lessons learned
-
Lesson Learned 1: Start with a strategy based on a sharded landscape and vision of the native digital manufacturing site.
-
Lesson Learned 2: Partner with quality early, define common operating procedures, clear testing methodology, and set guidelines to ensure compliance.
-
Lesson Learned 3: Integrate vendors into your culture. Most vendors want everything completely defined first before starting , but when you’re trying to do something innovative and new , this will not achieve the goal. Collaboratively working together and trying new things including some that failed actually improved results and meeting our timelines.